LOADING 0%
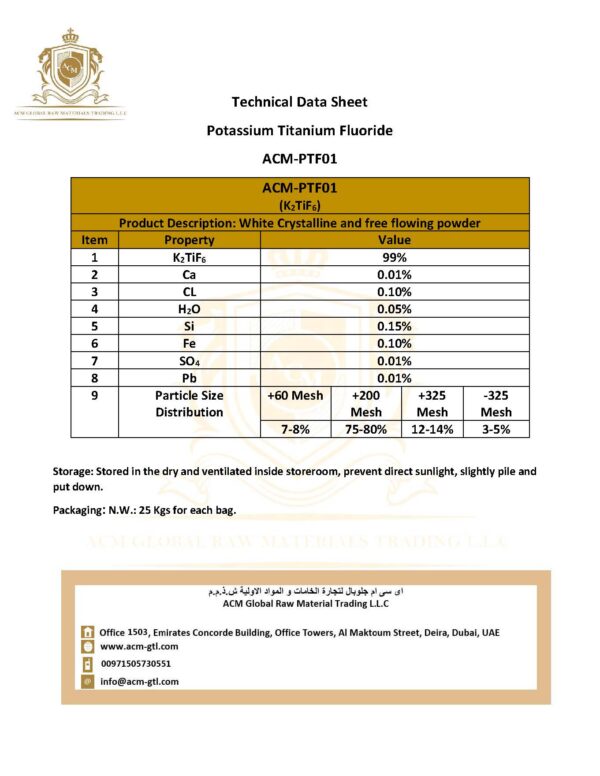
Potassium fluoride titanate is used in the production of titanium-boron-aluminum grain refining agents, boron-alloying tablets used for the manufacture of high quality aluminum alloys, and aluminum-boron hardeners. It is also used in textile processing, flux agents for casting, and as a component of flame retardants and fire preventing agents. Exposure can occur at either a potassium fluoride titanate manufacturing facility or at other manufacturing, packaging or storage facilities that handle potassium fluoride titanate. Persons involved in maintenance, sampling and testing activities, or in the loading and unloading of potassium fluoride titanate packages are at risk of exposure, but worker exposure can be controlled with the use of proper general mechanical ventilation and personal protective equipment. Although workplace exposure limits for potassium fluoride titanate have not been established, workplace exposure limits for fluoride ion (F-) should be used in worksite safety programs. Please see the MSDS for additional information. The general public and consumers are not expected to be exposed to potassium fluoride titanate. Potassium fluoride titanate is a white powder that is stable under normal conditions of storage and use. It is not compatible with oxidizing agents. Hazardous decomposition products formed under fire conditions include hydrogen fluoride, potassium oxides, and titanium oxides. Potassium fluoride titanate is irritating to the skin and severely irritating to the eyes. It may also cause an allergic skin reaction. Breathing potassium fluoride titanate dust can irritate the upper respiratory tract. If ingested, potassium fluoride titanate is toxic. Short-term exposure to high concentrations or long-term exposure to lower concentrations of potassium fluoride titanate may cause fluoride poisoning with stomach pain, weakness, convulsions, collapse and death. Long-term (chronic) exposure may also cause deposits of fluorides in bones and teeth, a condition called fluorosis, which may result in pain, disability and discoloration or mottling of teeth. Currently, there is no evidence that potassium fluoride titanate is a reproductive or developmental toxic substance. The cancer risk for potassium fluoride titanate is low. Potassium fluoride titanate is soluble in water and therefore will dissociate into its constituent ions in the aquatic environment. Potassium fluoride titanate may be toxic to aquatic organisms, which is essentially due to the fluoride ion (F-). Fluoride tends to accumulate preferentially in the skeletal and dental hard tissues of vertebrates (fish), exoskeletons of invertebrates, and cell walls of plants. No accumulation has been reported for edible tissues. For risk management, the World Health Organization (WHO) has recommended a guideline of 1.5 mg/L for fluoride in drinking water.